 Identify the compound/compounds which does not obey octet rule:Īns. Higher the bond order smaller will be the bond length.ġ5. Bond order is the number of bonds between two atoms while bond length is the equilibrium distance between the nuclei of two bonded atoms in a molecule. What is difference between bond order and bond length and state relation between them?Īns. Therefore, there is no way to arrange five pairs of bonding electrons around a nitrogen atom.ġ4. NCl 5 does not exist because there are no d-orbitals. PCl 5 forms five bonds by using the d-orbitals to “expand the octet” and have more places to put bonding pairs of electrons. A molecule of PCl 5 exists while that of NCl 5 does not.Īns. 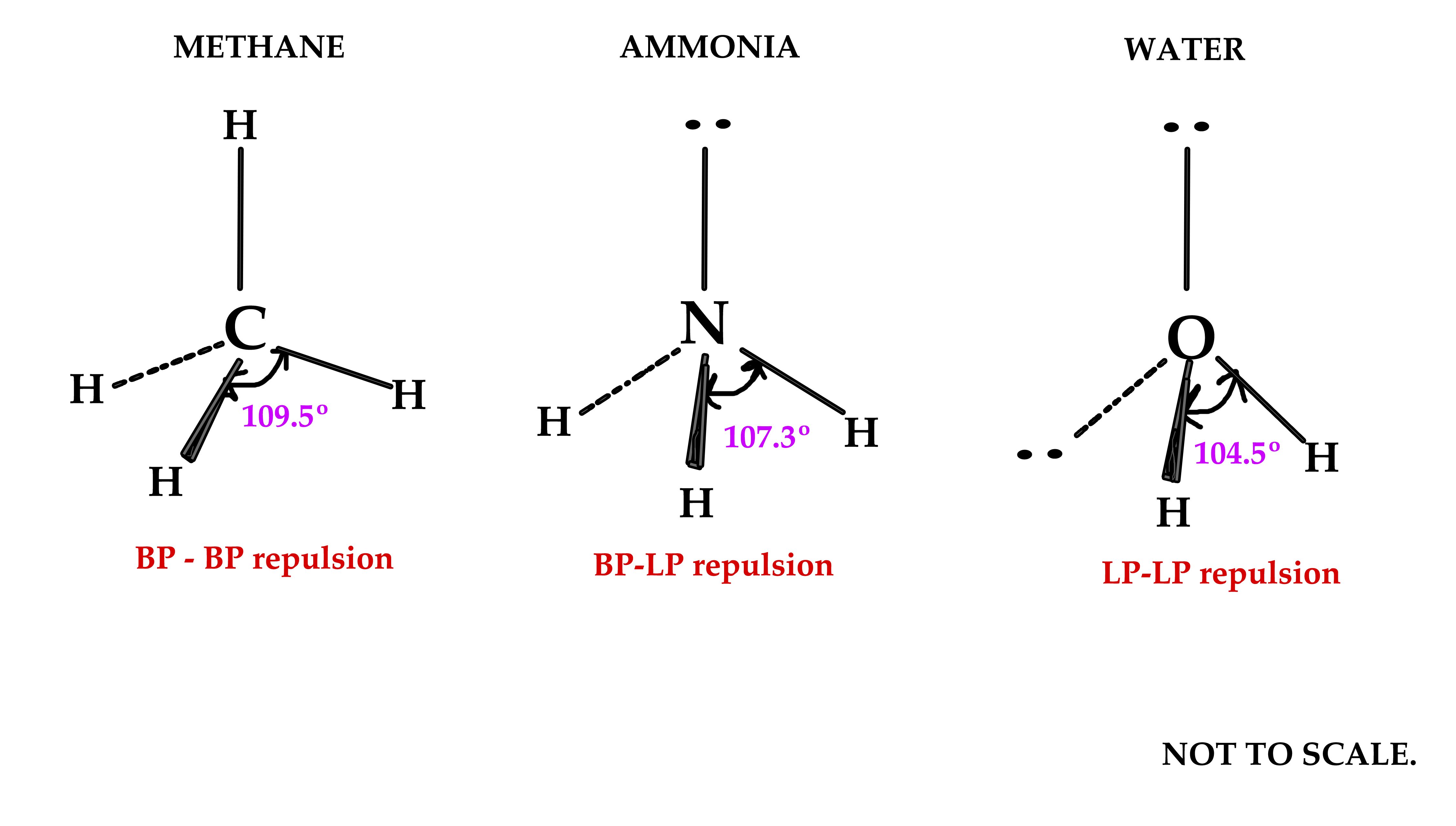
It is the property of an isolated atom.ġ3. Whereas electron gain enthalpy is the tendency of an atom to attract outside electron. Electronegativity: Electronegativity is the tendency of an atom to attract shared pair of electrons towards itself. How does it differ from electron gain enthalpy?Īns. A plus sign in an orbital indicates a positive wave function while a minus sign in an orbital represents a negative wave function.ġ2. Molecular orbitals are represented by wave functions. Write the significance of plus and minus sign shown in representing the orbitals.Īns. sp 3 : CH 4 sp 2 : C 2 H 4 sp: C 2 H 2ġ1. Name one compound each involving sp 3, sp 2, sp hybridization.Īns. Give two examples of molecules containing coordinate bonding.ġ0. Give one example each for a compound withĩ. In N 2 molecule energy of π2p x and π2p y are _ then σ2p z. The bond in peroxide ion is _ then in superoxide ion.(Weaker /Stronger)ĥ. Out of HF, HCl, HBr and HI, the lowest boiling point is of HCl and highest boiling point is of HF.Ĥ. Out of HF, HCl, HBr and HI, the lowest boiling point is of _ and highest boiling point is of _.Īns. In the solid state, Na + and Cl – ions are strongly held together and are not free to move.ģ. Why is NaCl a bad conductor of electricity in the solid state?Īns. False shape of IF 7 is pentagonal bipyramidalĢ. 
The shape of SF 6 molecule is octahedral whereas that of IF 7 is square pyramidal.Īns. 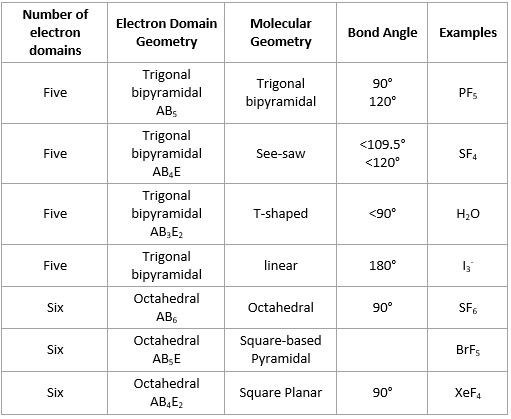
Revision notes become a crucial thing for those students who are actually serious about scoring good grades.ĭownload CBSE Class 11 Chemistry Revision Notes 2023-24 PDFĪlso, check CBSE Class 11 Chemistry revision notes for other chapters: Through chemical bonding Class 11 notes, it becomes easy for the students to know all the essential things that are present in the chapter. These notes are prepared by some expert Chemistry teachers. Revision notes of Chapter 4 Chemistry Class 11 can help students to get good marks in Class 11th examinations as they cover all the necessary topics, concepts and terms in a fun and simple language for the proper understanding of students.ĭuring the examination Class 11th Chemistry Chapter 4 notes about Chemical Bonding and Molecular Structures become very important. One such chapter is Chapter 4 - Chemical Bonding and Molecular Structures. There are many important, and easy chapters are there in Class 11 Chemistry which can easily help students attain good marks in exams. With proper guidance and with appropriate notes, good marks can easily be achieved in Chemistry in Class 11th. Students generally think Class 11th Chemistry is tough and good marks cannot be scored in this subject, but this doesn't seem right. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
